A51193EJ
INTERVENTIONS FOR VALVULAR DISEASE
2nd generation mitral posterior leaflet replacement
ULRICH S. (1), ALEX K. (2), FABIO S. D. B. J. (2), IRAKLI G. (3), JACQUES E. (4), ALEX K. (4)
(1) HGZ, Bad Bevensen GERMANY
(2) InCor - Instituto do Cora??o do Hospital das Cl?nicas da FMUSP, State of S?o Paulo BRAZIL
(3) Healthycore ????????? - ??????? - ??????????? ?????????? ???????? ???????, Tbilisi GEORGIA
(4) Polares Medical, CA UNITED STATES
(1) HGZ, Bad Bevensen GERMANY
(2) InCor - Instituto do Cora??o do Hospital das Cl?nicas da FMUSP, State of S?o Paulo BRAZIL
(3) Healthycore ????????? - ??????? - ??????????? ?????????? ???????? ???????, Tbilisi GEORGIA
(4) Polares Medical, CA UNITED STATES
Company
Polares Medical SA, a spin-off of Symetis SA?which was acquired by Boston Scientific in 2017?is developing the MRace, a mitral hemi-valve designed to address mitral regurgitation (MR). The device is currently in the early stages of clinical development. Polares has offices in Lausanne, Switzerland, and Palo Alto, USA.
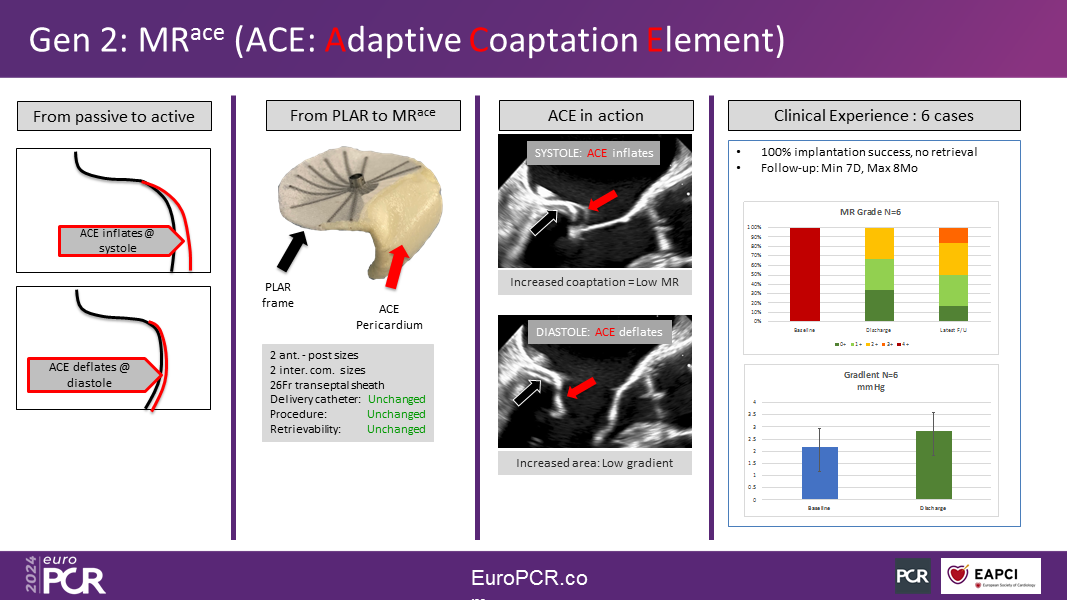
The MRace hemi-valve, replacing the posterior leaflet, aims to restore coaptation with the native anterior leaflet, thus reducing MR without creating a trans-mitral gradient. It is constructed from a pericardium membrane mounted onto a nitinol/ePTFE frame.
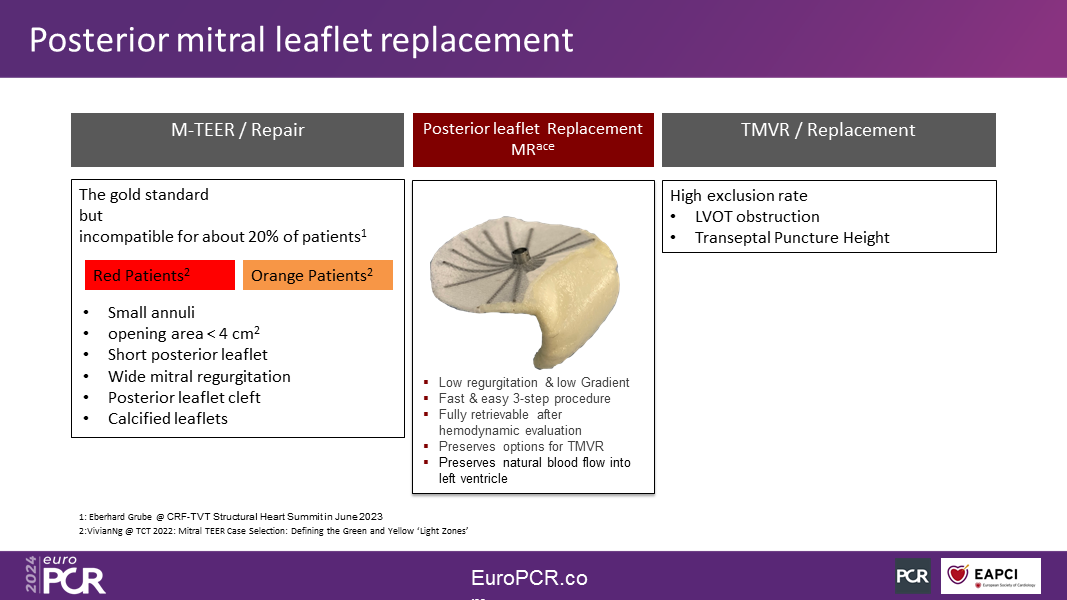
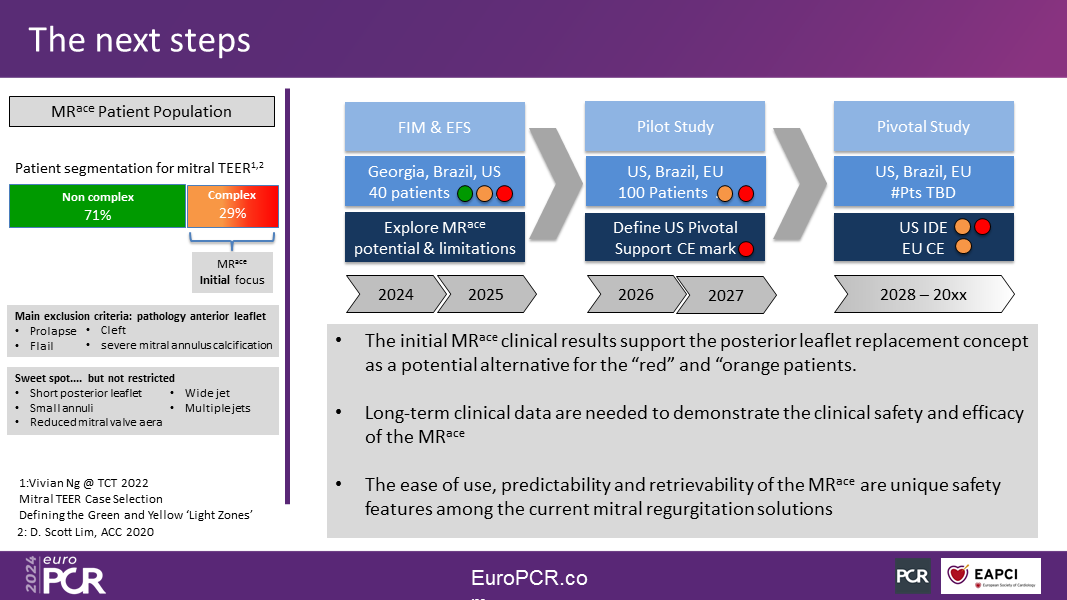
The initial clinical application of the MRace hemi-valve targets patients with complex mitral valve anatomies, which are known to pose challenges for edge-to-edge repair (TEER), such as a short posterior leaflet, small opening area, small annuli, wide MR, and posterior leaflet cleft.
However, the MRace offers unique advantages over TEER and transcatheter mitral valve replacement (TMVR). It is an easy, predictable, and fast procedure; it is fully retrievable after hemodynamic assessments (MR reduction, trans-mitral gradient) in case of an undesired functional outcome; and it preserves the option for future TMVR treatment if necessary.
Furthermore, since the anterior mitral leaflet is preserved, the MRace hemi-valve is expected to maintain the physiological vortex formation during diastole and prevent the energy loss typically observed after mitral valve replacement. This could lead to improved cardiac output and potentially longer-term benefits compared to TEER and TMVR, pending clinical outcomes that confirm its safety and efficacy.
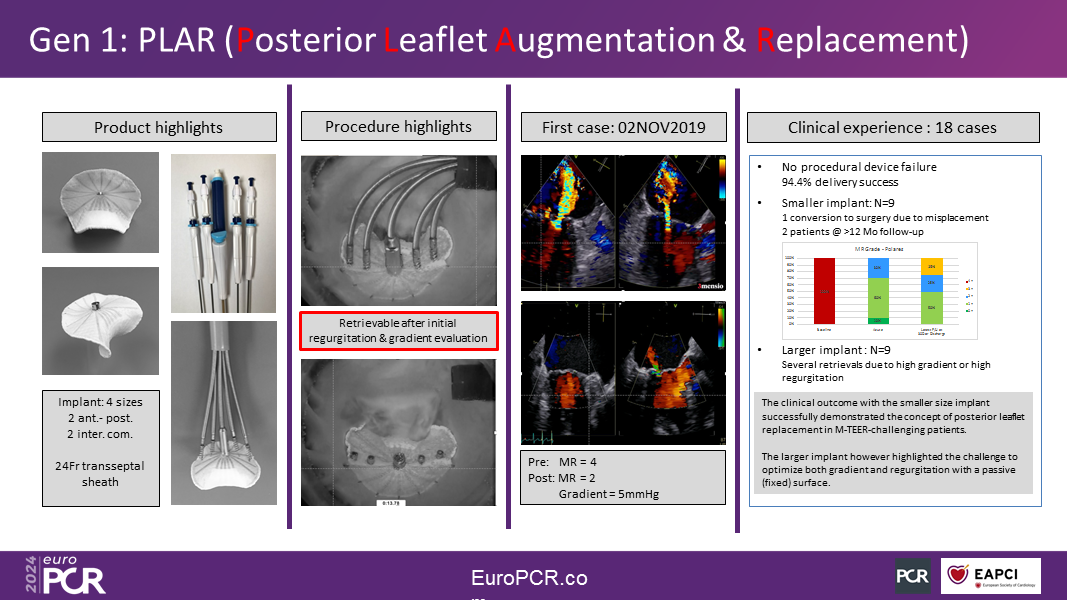
Polares? first-generation hemi-valve replacement was evaluated in an 18-patient clinical trial, which confirmed the concept, establishing safety and efficacy in reducing MR. This trial, however, highlighted the paradigm of reducing MR without increasing the trans-mitral gradient.
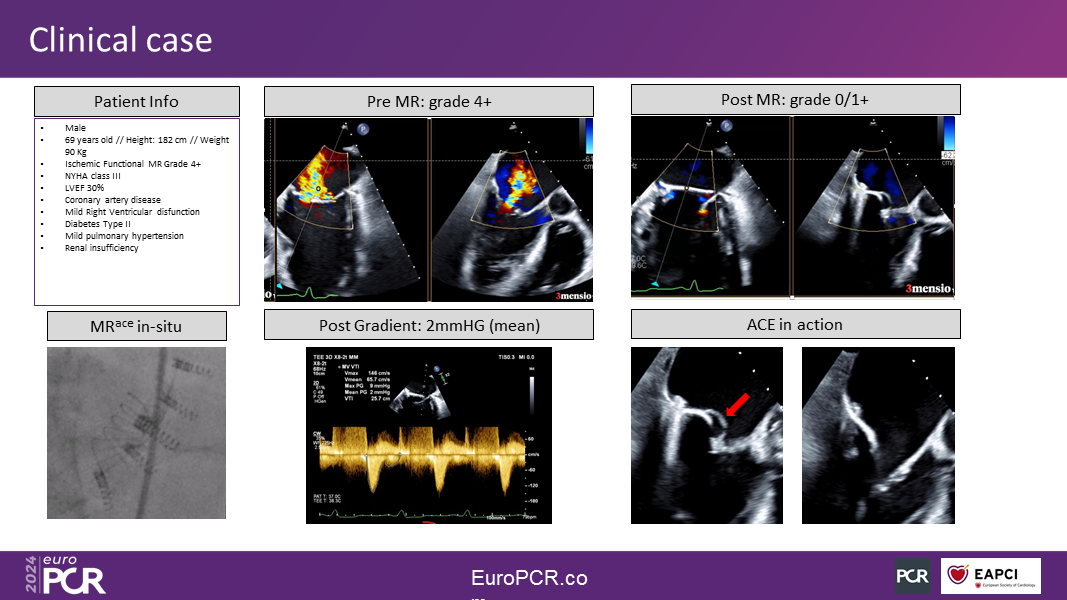
The second-generation MRace hemi-valve was recently implanted in eight patients under a First-in-Human clinical study protocol in Tbilisi, Georgia, and Asuncion, Paraguay. It reduced MR by an average of three grades while effectively maintaining the trans-mitral gradient unchanged. Parallel clinical studies will soon commence in Brazil, the USA, and Europe, with the initial goal to gather sufficient clinical experience to design and launch a large-scale (100 patients) pilot study in the US and other regions, focusing on complex mitral anatomies.
Polares has a robust intellectual property portfolio with early priority dates, pre-dating potential competitors. Its first patents, filed as early as 2011, cover key commercial aspects of the technology. Polares is supported by a strong syndicate of investors, including Decheng, Endeavour Vision, Longitude Capital, Wellington Partners, EarlyBird, and IDO.
For further contact:
Jacques Essinger, Ph.D, CEO
Email: jessinger@polaresmedical.com
The MRace hemi-valve, replacing the posterior leaflet, aims to restore coaptation with the native anterior leaflet, thus reducing MR without creating a trans-mitral gradient. It is constructed from a pericardium membrane mounted onto a nitinol/ePTFE frame.
The initial clinical application of the MRace hemi-valve targets patients with complex mitral valve anatomies, which are known to pose challenges for edge-to-edge repair (TEER), such as a short posterior leaflet, small opening area, small annuli, wide MR, and posterior leaflet cleft.
However, the MRace offers unique advantages over TEER and transcatheter mitral valve replacement (TMVR). It is an easy, predictable, and fast procedure; it is fully retrievable after hemodynamic assessments (MR reduction, trans-mitral gradient) in case of an undesired functional outcome; and it preserves the option for future TMVR treatment if necessary.
Furthermore, since the anterior mitral leaflet is preserved, the MRace hemi-valve is expected to maintain the physiological vortex formation during diastole and prevent the energy loss typically observed after mitral valve replacement. This could lead to improved cardiac output and potentially longer-term benefits compared to TEER and TMVR, pending clinical outcomes that confirm its safety and efficacy.
Polares? first-generation hemi-valve replacement was evaluated in an 18-patient clinical trial, which confirmed the concept, establishing safety and efficacy in reducing MR. This trial, however, highlighted the paradigm of reducing MR without increasing the trans-mitral gradient.
The second-generation MRace hemi-valve was recently implanted in eight patients under a First-in-Human clinical study protocol in Tbilisi, Georgia, and Asuncion, Paraguay. It reduced MR by an average of three grades while effectively maintaining the trans-mitral gradient unchanged. Parallel clinical studies will soon commence in Brazil, the USA, and Europe, with the initial goal to gather sufficient clinical experience to design and launch a large-scale (100 patients) pilot study in the US and other regions, focusing on complex mitral anatomies.
Polares has a robust intellectual property portfolio with early priority dates, pre-dating potential competitors. Its first patents, filed as early as 2011, cover key commercial aspects of the technology. Polares is supported by a strong syndicate of investors, including Decheng, Endeavour Vision, Longitude Capital, Wellington Partners, EarlyBird, and IDO.
For further contact:
Jacques Essinger, Ph.D, CEO
Email: jessinger@polaresmedical.com
Click on the stars to rate: